OUR PHILADELPHIA FACILITIES
Interested in a tour of our Philadelphia Lab?
Philadelphia Laboratories






Philadelphia Capabilities
On-Site Technologies
Micro-computed tomography (microCT) scanners
Optical microscopes
Surface interferometers
Fourier transform infrared (FTIR) spectrometer
Scanning electron microscope with dispersive X-ray spectrometer (SEM/EDS)
Single and multi-axial mechanical testing and measurement systems
Machine shop
3D printer
Related Evaluations

Biomedical Evaluations
Create advanced medical devices and solutions by working with pioneers in the field.

Mechanical Evaluations
Understand your product's strengths and limitations with customized mechanical testing.

Materials Evaluations
Build a strong foundation for your products with specialized materials evaluations.
![Battery Supply Chain Services [EECS]](/sites/default/files/styles/cards_home_card/public/media/images/GettyImages-1411358298.jpg.webp?itok=Yd7-744o)
Battery Evaluations
Gain confidence with our cutting-edge evaluations and consulting.
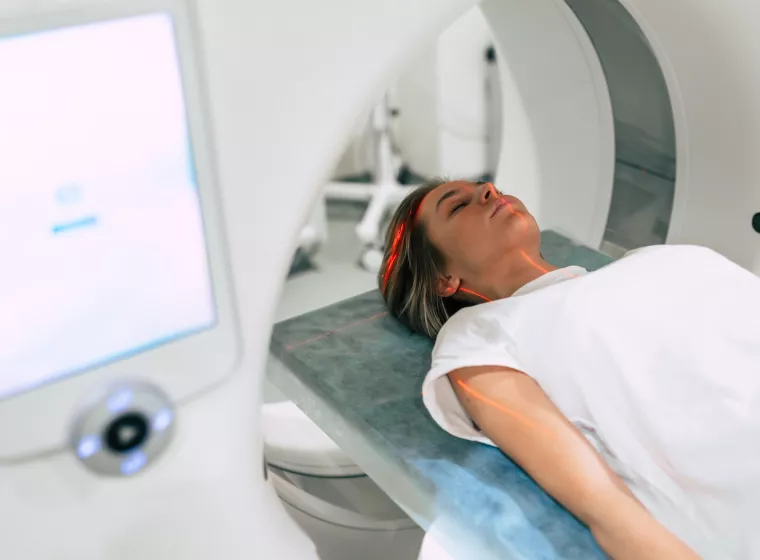
MRI Compatibility
MRI compatibility testing for medical devices to help ensure product safety and regulatory compliance.

Inform Your UX Decisions With Science
Our Exponent user experience (UX) teams design safe, successful user experience rooted in a deep understanding of your audiences. While we can tackle virtually any UX challenge, we offer particular expertise in automotive products, consumer products, industrial equipment, and medical devices.
Scope of Accreditation to ISO/IEC 17025:2017
Our Philadelphia Laboratory is accredited to ISO 17025 by A2LA (Certificate 2561.01) for a range of biomedical, biomaterials, biotribology, electrochemical, and spectroscopy tests and, as applicable, our services are compliant with 21 CFR Part 58 and U.S. FDA Good Laboratory Practices (GLP). Our lab is accredited to perform the following tests:

Best-in-Class Facilities Meet World-Class Experts
Our multidisciplinary teams of scientists and engineers at Exponent facilities in the U.S., Europe, and Asia are uniquely positioned to provide rapid response, onsite inspections, bespoke testing, and unparalleled scientific analysis, data, and research.